How ‘Idle’ Egg Cells Defend Their DNA From Damage
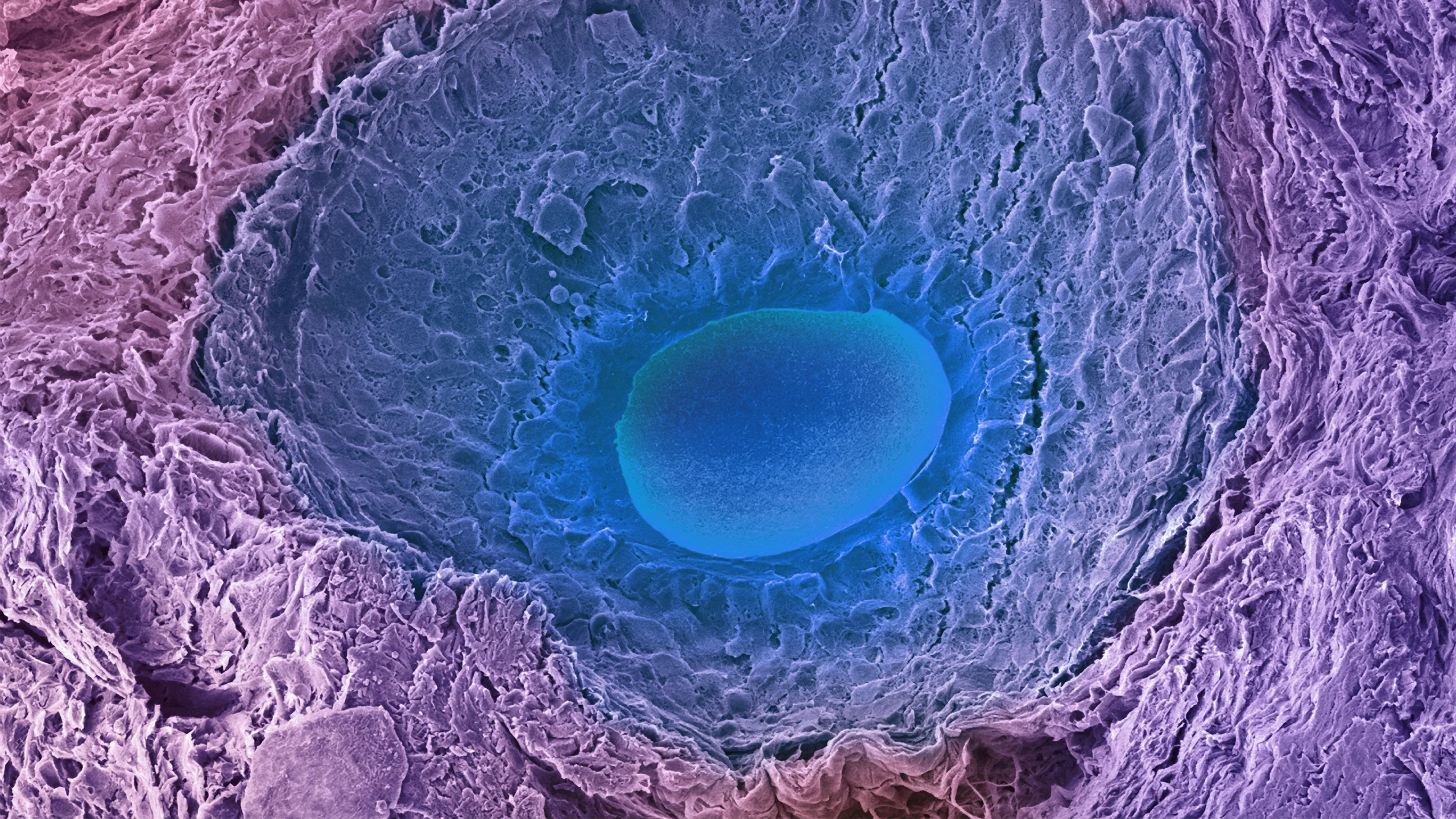
An oocyte, engulfed by the follicle and cells that support its decades-long life span, has an unusual way of dealing with contaminants to preserve its mitochondrial DNA and cytoplasm — both of which are passed on to the next generation.
Steve Gschmeissner/Science Source
Introduction
Out of all the cells in the body, oocytes are the most patient. The immature egg cells form inside a female’s body when she’s still a fetus in her mother’s womb, and then they wait in a quiescent state for years, if not decades. Cocooned inside ovaries, they pause, neither dividing nor replicating their DNA, so that one day they may pass along pristinely preserved genetic information to the next generation.
Yet sitting quietly for year upon year without degrading is immensely challenging for cells. Dormancy may reduce the wear and tear on oocytes, but it also reduces their ability to repair the inevitable damage they sustain at the molecular level. Recently, in a study published in Cell, researchers discovered how mouse oocytes keep DNA safe from damaging proteins. The cells construct special compartments within themselves to temporarily sequester the proteins. Then, heeding a molecular signal, the storage units disperse all at once, practically vanishing along with their dangerous cargo. Thus cleaned, the oocyte is left to mature safely.
The results are “beautiful and super interesting,” said Marie-Hélène Verlhac, a cell biologist at the Collège de France Center for Interdisciplinary Research in Biology, who was not involved in the study. They show how unusual oocytes are, she added. “You have the impression they’re unique.”
The work reveals a new way that oocytes quietly work to preserve genetic quality, and opens new avenues of research into addressing problems with fertility.
Oocytes are long-lived, an uncommon trait among cells. A female human baby is typically born with 1 million to 2 million of them. Early in life, the cells are tiny and generally don’t do much — researchers often call them “idle.” But over time they get much bigger: During the first decade of life, human oocytes grow 64-fold — a “massive expansion” that demands a lot from the energy-producing cellular organelles called mitochondria, said Elvan Böke, a cell biologist at the Center for Genomic Regulation in Barcelona, Spain. The number of mitochondria in oocytes balloons from just a few thousand to hundreds of thousands.
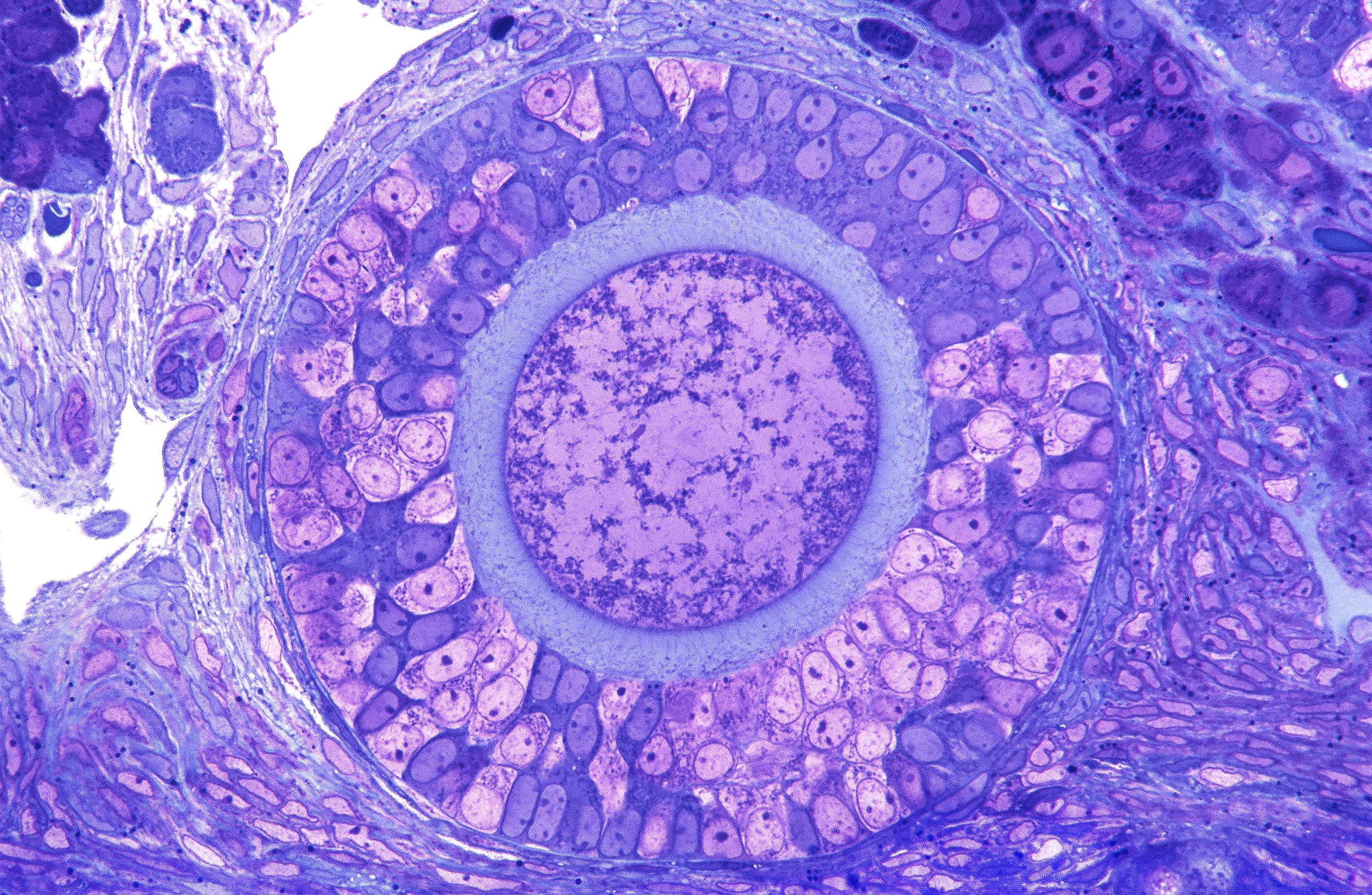
An oocyte rests in an ovarian follicle, where it remains dormant for years and is nurtured by surrounding granulosa cells. After a female goes through puberty, every month a hormonal signal triggers one oocyte out of hundreds of thousands to mature.
MICROSCAPE/Science Source
Having lots of mitochondria is normally risky for cells because their chemical activity generates toxic byproducts known as free radicals. But when Böke peered inside dormant human and frog oocytes, they weren’t overloaded with free radicals at all. As she reported in a Nature paper published in 2022, the oocytes’ mitochondria were doing something surprising: They were skipping the step in their energy-generating process that produces these dangerous molecules. This meant that they produced less energy overall; Böke speculated that they direct all the energy they do produce for growth. No other cell had been observed to do this.
“Oocytes are crazy,” said David Pépin, associate director of the pediatric surgical research labs at Massachusetts General Hospital. “They have more mitochondria than any other cell, and those mitochondria are weird.”
The perils for long-lived cells go beyond free radicals. Scientists have seen how energy production and growth generate many proteins, including clumps of misfolded and damaged ones. To clear this harmful detritus, neurons, which like oocytes are long-lasting cells that don’t divide, rely on biochemical pathways that refold and degrade bungled proteins. When this machinery fails, protein aggregates can devastate entire neural networks, snuffing out communication and leading to neurodegenerative diseases like Alzheimer’s, Parkinson’s, Huntington’s and ALS.
After Böke published her 2022 research on free radicals, she wondered how oocytes tamp down problematic proteins. She dug through thousands of papers but couldn’t find any studies characterizing how oocytes cope. So, she looked to mouse oocytes to find the answer.
Because oocytes expand so massively and produce proteins so intensively during their growth period, Böke expected to see high protein activity in the two major pathways responsible for clearing aggregates. Instead, she saw less activity in immature oocytes than in mature egg cells.
“It doesn’t make much sense,” she said. “Why would you put your degradative activity down if you’re going to grow and make lots of things?”
It was because the oocytes weren’t degrading the damaging proteins — they were storing them to destroy later.
When Böke and her team infused oocytes with a dye that illuminates protein aggregates, she saw the proteins clustered in large compartments. Probing further, she used electron microscopy to reveal these distinct storage units as super-organelles, which she named ELVAs, or endolysosomal vesicular assemblies. These pouchlike objects are composed of lysosomes — the cell’s waste-disposal organelles — and other protein-degrading machinery.
In young oocytes, they are situated throughout the cytoplasm. Later, as oocytes mature, ELVAs fuse together, relocate and ramp up their degradative machinery. By the time an oocyte is fully mature, harmful protein aggregates are undetectable, and the ELVAs vanish, revealing a clean cytoplasm.
It’s possible that other long-lived, nondividing cells have similar machinery that uses this catch-and-release strategy for disposing of cellular waste. For example, researchers have found that some long-lived stem cells also have ELVA-like components, which capture and store aggregates until the cells are ready to divide. Neurons, too, can develop triage centers, called aggresomes, for organizing misfolded proteins. However, neurons destroy their cellular detritus throughout their life spans, while oocytes store them and then wipe them out all at once. Böke speculated that the oocytes’ method is more energy-efficient: Since their mitochondria produce less energy overall, they need all of it for necessary growth to reach reproductive maturity.
Böke’s findings offer new details and a fresh perspective on the biology of oocytes. They also suggest new approaches to fertility research. Clinical research has shown that older people are less likely to get pregnant than younger ones. The standard explanation is that their eggs may be “too old” for pregnancy. But what if an old egg is simply one that has accumulated too many misfolded proteins?
Böke’s lab is currently collaborating with fertility clinics to look at protein aggregates in human oocytes. She suspects that elements related to oocyte growth and quality control, such as mitochondria and protein aggregation, may account for some unexplained infertility cases. “A broken system gives errors in many different ways,” she said. The key to identifying those errors, and addressing them, will likely come from research, like Böke’s, that seeks to understand fundamentally how oocytes work.



